Bacterial Translocation in Liver Cirrhosis
Lorraine Cunningham, Alan O’Sullivan-Weekes & Sarah Ward
Contents
Introduction
Bacterial translocation (BT) can be defined as the passage of bacterial flora and microbial products from the intestinal lumen to mesenteric lymph nodes (MLNs) or other extra-intestinal sites (Merli et al, 2015). Several studies have demonstrated the importance of BT in cirrhosis and it’s role in the pathogenesis of spontaneous bacterial peritonitis (SBP) (Runyon et al, 1994). Although the mechanisms driving the translocation of the gut flora in cirrhosis are not fully understood, recent evidence suggests that translocation of bacteria and bacterial products, such as endotoxin from the intestinal lumen into the systemic circulation, is a key factor in the pathogenesis of chronic liver diseases and the development of complications in cirrhosis (Pijls et al 2013). In addition to changes in the intestinal flora and the immune system, dysfunction of the intestinal epithelial barrier may also contribute to BT (Pijls et al, 2013). This review discusses the multiple mechanisms involved in the process of BT, with an emphasis on alterations in intestinal flora and mucosal barrier function (Wiest et al, 2014).
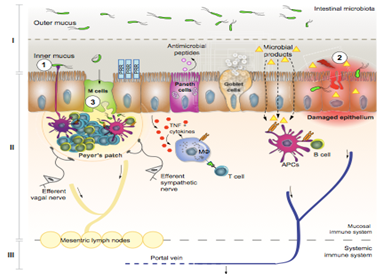
Compartments involved in pathological bacterial translocation
(Wiest et al, 2014)
|
Adapted with permission from (Wiest et al, 2014)
GALT
Gut associated lymphatic tissue (GALT) is a defence organ against bacteria. The bacteria microbial-associated molecular patterns (MAMPs) are recognised by pattern recognition receptors (PRR). In response to BT, gut epithelial cells release chemokines. This will attract dendrite cells (DCs) to the mucosa. DCs induce production and maturation of B cells and T cells which are then released in to the blood. In cirrhotic patients, a reduction of B cells can be seen. The degree to which this contributes to BT is currently unknown. Reduction of T cells causes an accumulation of bacteria in the MLN and the spreading of bacteria to extra intestinal sites (Wiest et al, 2014).
MLN
Part of the GALT and play a role in the adaptive and the innate immune response (Bellot et al, 2013). Normally bacteria transported to MLN by DCs are killed without inducing systemic immunity. Immune-suppression can permit the translocation of bacteria from intestines, which can lead to sepsis and death (Wiest et al, 2014). Patients with cirrhosis have systemic immune alterations which may promote development of infections and BT. Examples of these alterations include: decreased activity of reticuloendothelial system (RES) which are a defence against bacteraemia and other haematogenous infections. This will have a marked effect on bacterial products such as endotoxin or bacterial DNA (Bellot et al, 2013). Advanced cirrhosis is associated with a decrease in both cellular and humoral components of the immune response (Bellot et al, 2013).
Mechanical components
The epithelium facilitates absorption of luminal nutrients, water and electrolytes. It also acts as a barrier to prevent translocation of potentially harmful substances via transcellular and paracellular transport ( see an extensive review of the literature, Pijls et al 2013). The mucosal epithelium plays an important role in the prevention of infiltration of bacteria into the host as seen in figure 1. (Wiest et al, 2014). Structural abnormalities in the intestinal wall show an increased potential for BT in cirrhosis (Riordan and williams, 2006). Although, little investigation has been carried out in relation to cirrhosis (Wiest et al, 2014).
Secretory components
MAMPs secreted by the paneth cells are directly linked to bacteria and lipopolysaccharide (LPS) exposure. These play a role in the balance of the intestinal host microbial interphase (Wiest et al, 2014). Paneth cells are located at the bottom of each intestinal crypt. MAMPs secreted include: alpha-defenins in response to bacteria and LPS; Lysozme and secondary phospholipase A2 involved in local defence against bacteria; Beta-defensins antimicrobial peptides are expressed by most epithelial cells of the intestines (Tsao et al, 2005).
Mucus
This prevents direct contact of bacteria with the microvilli. In recent studies on alcoholics a thickness of the mucosa in the duodenum can be seen. This indicates a change in relation to cirrhosis or alcohol. An absence of bile has been shown to increase BT. In cirrhosis a decrease in the concentration of bile salts can be observed leading to an increase in BT (Wiest et al, 2014). In a study rats with cirrhosis were given conjugated bile acids which led to reversal of small intestinal bacterial overgrowth (SIBO), lowering the rate of BT and thereby improving the survival chances (Riordan and Williams, 2006).
IgA Antibodies
Secreted into the gut lumen where it binds and aggregates bacteria. IgA also neutralise toxins. A reduced amount of igA wil cause an increase in BT. In cirrhotic patients a decrease of fecal igA concentration and decreased secretion on igA can be observed. This may show a link between igA, BT and development of infections in cirrhosis but this is yet to be proven (Wiest et al, 2014).
Bacterial over-growth
Small intestinal bacterial overgrowth (SIBO) is an increased number and/or abnormal type of bacteria in the small intestines. This is one of the main factors promoting BT. it has been shown that cirrhotic rats with ascites and BT have a higher rate of SIBO compared to those without ascites. Studies have also revealed that SIBO is more common in patients with cirrhosis than in controls, especially in patients with advanced liver dysfunction and that have a history of SBP (Bellot et al, 2013). Increased fecal counts of both gram-positive and gram-negative bacteria species have been reported when cirrhosis is present, SIBO has also been demonstrated. (Riordan and williams, 2006).
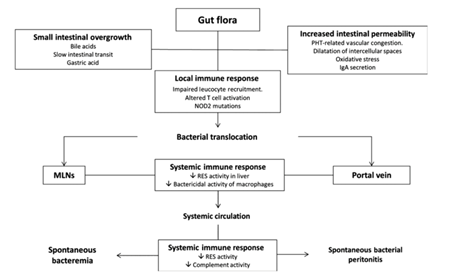
Pathophysiology of BT
Making studies using experimental models is the main way in which we study pathogenesis of the BT. This is due to the difficulty in accessing MLNs in patients with cirrhosis. In experimental models, BT is defined as a MLN-positive culture, 56% of rats with CCl4-induced cirrhosis and ascites had BT but in animals without ascites it drops to more like 0-10%. Patients with cirrhosis and BT had a higher degree of liver dysfunction in comparison to patients without BT. (Bellot et al, 2013) Ascites from the Greek word meaning “baglike” is a gastroenterological term for an accumulation of fluid in the around the stomach area. The medical condition is also called peritoneal cavity fluid and is most usually due to cirrhosis. (Pedersen et al, 2015) BT was investigated at two stages of experimental portal hypertension in another study, acute (meaning when shunting is minimal) and chronic (meaning when shunting is extensive and is like the portal hypertension of cirrhosis). In this study they found that BT is a command event in the acute portal hypertension model most likely because of a greater mesenteric inflammation. However, no difference in the rate of BT in chronic portal hypertensive was detected in rats (15 days after portal vein ligation) compared with control rats. It has been noticed that patients with cirrhosis and ascites and the presence of bacterial DNA in serum, a surrogate marker of BT, had the same degree of portal hypertension as patients without bacterial DNA. (Bellot et al, 2013) There are at least three mechanisms shown in figure 2, that influence the pathogenesis of BT which are: (Bellot et al, 2013)
• Small intestinal bacterial overgrowth
• Increased intestinal permeability
• Immune alterations of cirrhosis
|
Adapted with permission from: (Bellot et al, 2013)
Small intestinal bacterial overgrowth (SIBO)
SIBO is a mechanism of excessive bacterial growth in the small intestine. (Quigley, et al, 2006). While certain research recommends microbial investigation of jejunal in the diagnosis, there are limitations that make its application difficult in clinical practice. It is an invasive test, in that microbial investigation places high demands on the quality of laboratory work. More than one test is needed to detect the distribution of bacterial overgrowth. One of the main factors contribulting to BT is likely linked to slowed intestinal transit, low acid gastric secretion, intestinal immunological factors and pancreatic and biliary secretions i.e. SIBO. In mice a direct relationship has been shown between numbers of a specific bacterial strain populating a segment of the intestine and numbers of bacteria of this strain present in MLN (Bellot et al, 2013).
Increased intestinal permeability
It is normal for the intestine to exhibit some permeability, for nutrients to enter and to have a barrier function to keep potentially harmful substances out (Campieri et al, 2002). The intestinal membrane consists of a mucinous component secreted by intestinal epithelial cells and intestinal epithelium, which forms a layer with intercellular junctions with selective passage of substances. Studies of cirrhosis have shown structural and functional alterations in the intestinal mucosa that increase intestinal permeability to bacteria and its products. However, it is not known if these structural changes are caused by BT. The majority of studies detailing intestinal permeability are performed using probes that measure paracellular permeability. However, BT of living bacteria has a transcellular route. Therefore, the studies of intestinal permeability may lack clinical accuracy (Bellot et al, 2013).
Immune alterations of cirrhosis
The intestinal tract consists of the GALT, the largest immunological organ of the body. It is comprised of MLN, lamina propria and peyers patches, which block BT as shown in figure 3. Otherwise BT may lead to bacteraemia or infections, particularly in cases of cirrhosis where there is already a reduction in immune efficiency (Bellot et al, 2013). Translocation of bacterial products promotes a chronic inflammatory response and impairs the haemodynamic changes observed in cirrhosis. From a different perspective, it has been demonstrated that administration of anti-TNF monoclonal antibodies to cirrhotic rats with ascites was associated with a significant decrease in the rate of BT. This means that the inflammatory response induced by BT also acts on the intestinal barrier permeability favouring the translocation of bacterial products (Bellot et al, 2013).
|
Adapted with permission from: (Wiest et al, 2014)
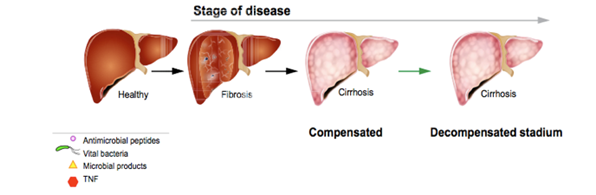
Stage of disease, route and site of pathological bacterial translocation
|
Adapted with permission from: (Wiest et al, 2014)
Stage of disease
The development of pathological BT increases with stage of liver disease seen in figure 4. Pathological translocation of bacteria to mesenteric lymph nodes is associated with the decompensated stage of liver cirrhosis. In a study carried out among cirrhotic patients, level of portal hypertension and liver insufficiency are the driving forces for BT (Reiberger et al, 2013). The latter appears to be the main culprit, as without liver insufficiency, chronic pre-hepatic portal hypertension does not lead to pathological BT (Kasravi et al,1996). Studies show the detection of small intestinal bacterial overgrowth SIBO in cirrhotic patients ranges from 48% to 73%. It has also been shown that it is more frequent in patients with more severe liver disease (Wiest et al, 2014).
Site of bacterial translocation
The colon harbours trillions of bacteria and serves as an effective machine in eliminating translocating bacteria (Powell, 1981). Experimental studies have revealed that after introducing equal concentrations of E. coli into small or large bowel, BT occurs at a greater rate following small bowel inoculation(Koh et al, 1996). In addition, transit time of bacterial translocation at the level of the mucosa and muscularis were significantly enhanced in animals with developing cirrhosis compared to controls (Palma et al 2007).
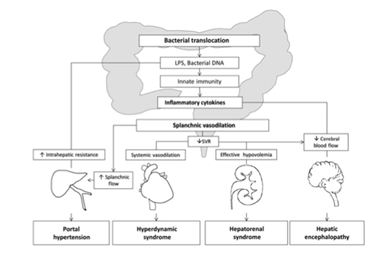
Clinical Importance of bacterial translocation
Intestinal bacterial overgrowth and increased BT of gut flora from the intestinal lumen leaves patients more susceptible to bacterial infections, major complications and also play a role in the pathogenesis of chronic liver disorders. Levels of bacterial LPS, a component of gram-negative bacteria, are increased in the portal systemic circulation in many different types of chronic liver disease, this can be seen in figure 5. (Ilan, 2012). Bacteria have been documented to migrate from the gut to systemic organs via two main routes; in the lymphatic or in the portal venous system. (Mainous et al, 1991). Bacteria can be detected earlier in the portal circulation and to a much greater extent than in the lymphatic system. This points towards the significance of hematological spreading of bacteria after crossing the epithelial barrier in cirrhosis. comparative and kinetic studies comparing the lymphatic and the portal-venous routes in BT relating to liver cirrhosis are not currently available (Wiest et al, 2014). Types of membrane proteins called Toll-like receptors (TLR) are involved in innate immune responses, which recognize various molecular patterns of pathogens. The activation of these receptors is followed by the release of pro-inflammatory cytokines that can worsen the haemodynamic changes of cirrhosis, especially splanchnic vasodilation and hyperdynamic syndrome. TLR agonists have been shown to induce tumour necrosis factor (TNF) secretion by monocytes. BT can cause the hepatic and systemic haemodynamic abnormalities of liver cirrhosis. (Bellot et al, 2013) Studies in cirrhotic rats with BT have an increased activity of endothelial nitric oxide synthase (eNOS) in mesenteric blood supply. Studies in humans have shown that cirrhotic patients with increased levels of LBP, an indirect marker of BT, have a more profound systemic haemodynamic derangement. It has been suggested that the worsening of the hyperdynamic circulation and the inflammatory state associated with BT may play a role in the complications of portal hypertension. Kidneys with cirrhosis have shown an increased expression of TLR4, and the pro-inflammatory cytokine TNF-a, which makes them susceptible to a further inflammatation during BT (Bellot et al, 2013). The most common infections seen in cirrhotic patients are Spontaneous Bacterial Peritonitis (SBP), Urinary Tract Infections and pneumonia, which are mostly caused by gram-negative bacilli (GNB), especially E.coli. This suggests most infections in these patients are of enteric origin. BT has been strongly linked to hepatic encephalopathy in patients with cirrhosis (Bellot et al, 2013).
|
Adapted with permission from: (Bellot et al, 2013)
Conclusion
BT is typically a healthy phenomenon. In certain circumstances, notably in decompensated stage liver cirrhosis, it is pathologically increased (Wiest et al, 2014). As previously discussed, a number of key factors are involved in promoting pathological BT in cirrhosis. These include microbiota (SIBO), the intestinal barrier (failure of mechanical barrier and secretory function) and the gut-associated lymphatic tissue (GALT) generating an immune response to eliminate sinister bacteria. Further drivers of pathological BT in cirrhosis include genetic susceptibility, malnutrition, pro-inflammatory cytokines, sympathetic hyperactivity (e.g., noradrenalin), and lack of bile acids (Wiest et al, 2014).
References
Bellot, P., Francés, R., Such, J. (2013). pathological bacteria translocation in cirrhosis: pathophiological diagnosis and clinical implications. Liver international , 33, (1), 31-39.
Campieri, M., Fiocchi, C., Hanauer, S. B. (2002). Inflammatory Bowel Disease: A Clinical Case Approach to Pathophysiology, Diagnosis, and Treatment. Springer , 7.
E., Quigley, R., Quera, Quera (2006). Small intestinal bacterial overgrowth: roles of antibiotics, prebiotics and probiotics. Gasteroenterology , 130, 78-90.
Garcia-Tsao, Wiest, R., Guadalupe. (2005). Bacterial Translocation (BT) in Cirrhosis. Hepatology , 41, (3), 422-433.
Koh IH, Guatelli R, Montero EF, Keller R, Silva MH, Goldenberg S (1996). Where is the site of bacterial translocation–small or large bowel? Transplantation Proceedings, 28, (5), 2661-2661.
Ilan, y. (2012). Leaky gut and the liver: a role for bacterial translocation in nonalcoholic steatohepatitis. World Journal of gastroenterology , 18 (21), 2609-2618.
JS., Pedersen; F., Bendtsen; S., Møller. (2015). Management of cirrhoic ascites. Therapeutic advances in chronic diseases, 6 (3), 124-137.
Kasravi, F. B., Wang, L., Wang, X., Molin, G., Bengmark, S., Jeppsson, B. (1996). Bacterial translocation in acute liver injury induced by D- galactosamine. Hepatology , 23 (1), 97-103.
Riordan, S.M, Williams, R., (2006). The intestinal flora and bacterial infection in cirrhosis. Journal of Hepatology , 45, 744-757.
Mainous, M., Tso, P., Berg, R., EA. Deitch. (1991). Studies of the route, magnitude, and time course of bacterial translocation in a model systemic inflammation. Archives of Surgery , 126 (1), 33-37.
Merli, M., Lucidi, C., Gregorio, V. D., Giannelli, V., Giusto, M., Ceccarelli, G. (2015). The chronic use of beta-blockers and proton pump inhibitors may affect the rate of bacterial infections in cirrhosis. Liver International , 35, (2), 362-369.
Palma, P., Mihaljevic, N., Hasenberg, T., Keese, M., Koeppel, T. A. (2007). Intestinal barrier dysfunction in developing liver cirrhosis: An in vivo analysis of bacterial translocation. Hepatology Research , 37, 6-12.
Pijls, K. E., Jonkers, D. M., Elamin, E. E., Masclee, A. A., Koek, G. H. (2013). Intestinal epithelial barrier function in liver cirrhosis: an extensive review of the literature. Liver International , 33, 1457-1469.
Powell, D. (1981). Barrier function of epithelia. American Journal of Physiology (241), G275–G288.
Runyon, B., Squier, S., Borzio, M. (1994). Translocation of gut bacteria in rats with cirrhosis to mesenteric lymph nodes partially explains the patho- genesis of spontaneous bacterial peritonitis. Journal of Hepatology , 21, 792-796.
Reiberger T, Ferlitsch A, Payer BA, Mandorfer M, Heinisch BB, Hayden H (2013). Non-selective betablocker therapy decreases intestinal permeability and serum levels of LBP and IL-6 in patients with cirrhosis. Journal of Hepatology , 58, 911-921.
Wiest, R., Lawson, M., Geuking, M. (2014). Pathological bacterial translocation in liver cirrhosis. Journal of Hepatology , 60, 197-209.