|
Size: 4640
Comment:
|
Size: 9895
Comment:
|
| Deletions are marked like this. | Additions are marked like this. |
| Line 110: | Line 110: |
{{attachment:food intake.jpg|alt text|height="400"}} == Molecular aspects of tanning == Tanning is understood as an increase in epidermal melanisation in skin following UV radiation, but can have a further role in a host response that protects against further UV- induced damage. Tanning is mainly seen in humans but has been known to be seen in other mammals such as sharks. (Barbara A. Gilchrest, 2011 ) Melanocytes (MC) sit along the epidermal basal layer, have a minimal role except for constitutive melanin production. They are usually only activated after sun exposure. (Graeme Walker et al, 2008) Coat color study in animals has been an important factor in the study of genes that control pigmentation in humans. In both humans and animals fully differentiated MCs (Melanocytes) can be divided into two major groups, those in the hair bulb, whose function is mainly to produce pigment for hair color, and epidermal MCs, which supply the epidermis with melanin and are involved in the tanning response. (Graeme Walker et al, 2008) After an UVR exposure to the skin, basal keratinocytes proliferate (resulting in epidermal hyperplasia), to replace apoptotic “sunburn cells” that express p53. (Graeme Walker et al, 2008) UVR may cause pigmentation in two ways by affecting the skin melanocytes. The first which can be called Intermediate pigment darkening, fades rapidly and is due to oxidation of already existing melanin in the melanocytes. The second, called Delayed tanning, appears a lot later as it increases melanogenesis, allowing the darkened skin to be evident a few days after UVR exposure and may last up to a week. (Graeme Walker et al, 2008) For a long time it has been appreciated that tanning is the skins main response in protection against UV radiation, and against acute and chronic damage. What we would consider a “good tan” at best offers a moderate degree of photoprotection. If we compare a lightly pigmentented epidermis with darkly pigemted epidermis, the darkly pigmented epidermis reduces skin UV damage by a factor of 4. Skin darkening regulated by UV radiation is a biphasic process. The initial darkening is due to redistributioing and molecular changes to already existing melanin pigments within epidermis. The second phase is due to up-regulation in melanin synthesis and its transfer to keratinocytes. (John D’Orazio et al, 2013) This can show that a number of changes occur in the epidermis due to the tanning response including melanogenesis, which is the synthesis of melanin – pigment in hair, eyes and skin color. (Barbara A. Gilchres, 2011) It is known that P53 Factor and KITLG expression are expressed in keratinized cells are exposure to UV. (Graeme Walker et al, 2008) It has been possible to prove that after 24 hours there is a 2-3 fold increase of nucleotide repair protiens, after UV radiation. Also research has discovered that keratinocyte derived gene products are regulated by UV. (Barbara A. Gilchrest, 2011) Researchers have been unable to discover how P53 regulates KITLG, but they have ideas based on the fact that most paracrine released from keritized cells after UV exposure are regulated by P53, and that KITLG has receptors for P53 allowing regulation. (Graeme Walker et al, 2008) UV has many other effects on the skin, including induction of an immune- tolerant or immunosuppressive state and production of vitamin D by direct conversion of 7- dehydrocholesterol into vitamin D3. (John D’Orazio et al,2013) == Artificial tanning == Over the years the number of artificial tanning methods such as indoor tanning salons have become more popular. In 1980 only 1% of the American population had ever used a sunbed, whereas now a huge 25% have used, or use them regularly. Indoor tanning salons are known to be poorly regulated in terms of the strength of UV radiation, they can be up to 10 times stronger than the UV inflicted by the sun, making the tanning beds an authentic carcinogenic instrument. It has been proven that tanning can be addictive, leading to more frequent and intense UV exposure. Lifetime risk of skin melanoma increases by 75%for sunbed users under the age of 35. Since the molecular pathways in the skin that activate UV-induced tanning result from cellular and DNA damage it can result in skin damage and carcinogenesis, it appears this is the same case for tanning salons but appears to be more intense. For business purposes the industry commercializes the UV health benefits, emphasizing the Vitamin D production, naturally made by the skin through 7-dehydrocholesterol into Vitamin D3, but research has suggested that every day sun exposure as well as Vitamin D containing foods and supplements exceed the required vitamin D levels the body needs. This means extra exposure through tanning salons is unnecessary to prevent diseases such as rickets. Decreasing UV radiation exposure, both naturally from sunlight and artificially from tanning bed use, may be the single best way to reduce incidence of melanoma and other skin cancers. (John D’Orazio et al, 2013) |
P53: The Key to Sunbathing (The Regulation of MSH)
Introduction to P53
This essay discusses the effects of P53, its relation to sunbathing and the tanning response.
Additionally, we will be discussing the P53’s relation to cancer and its prognostic factor and
the role of MSH.
Fig 1.
The Structure of P53 (Splettstoesser)
P53 (TP 53) is a tumor suppressor gene that is considered one of the most frequent targets
for gene alterations relating to cancer. (Moshe Oren et al, 2007) It codes for a protein that
regulates the cell cycle, functioning as a tumor suppressor. In 1993 P53 was voted molecule
of the year by science magazine, and has been described as “the guardian of the genome”,
relating to ability to prevent genome mutation.
In over half of human tumors we can see the inactivation of P53. Due to many experiments
on the biochemistry an physiology of P53 relating to its cancer role, it is now appreciated
that its main role is to prevent the emergence of cells with permanently defective genomes,
which are very likely to lead to cancer.
In normal cells, P53 protein is low, but after DNA damage or other stress such as UV
radiation, a trigger response may occur, increasing the number of P53 proteins in the cell.
These proteins have three major functions: apoptosis (cell death), DNA repair and growth
arrest – stopping the cell cycle and therefore preventing replication of damaged DNA.
Apoptosis would be a final result to prevent replication of damaged DNA and avoiding
tumor growth. Although high levels of P53 can be considered a good thing when referred to
cancer suppression, too much can result in accelerated aging which will be discussed further
as an effect of excessive UV radiation.
Role of P-53 and relation to MSH
P53 is a tumour suppressor gene and is located on the seventeenth (17p13) chromosome. It
is one of the most frequent targets for genetic alteration in cancer. A lot of human tumours
are able to mutationally inactivate p53, removing its role as a tumour suppressor and
preventing the emergence of cells with permanently defective genomes, which can often
cause cancer. P53’s ability is largely due to it being able to act as a sequence special c
transcriptional regulator. (Moshe Oren et al, 2007)
It is also a key regulator of cellular responses to geno-toxic stress, and it is stabilised and
activated after DNA damage. The rapid activation of p53 by ionising radiation mostly
dependents on the ATM kinase. P53 is phosphorylated by ATM shortly after DNA damage,
resulting in enhanced stability and activity of p53.
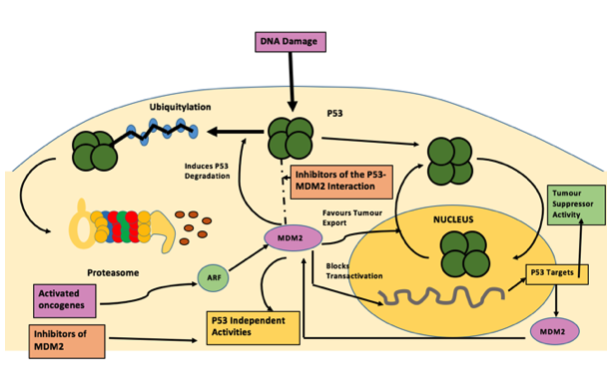
The Mdm2 oncoprotein is a major negative regulator of P53, triggering its degradation by
ubiquitin system. In response to ionising radiation and radio-mimetic drugs, Mdm2
undergoes rapid ATM-dependent phosphorylation before to p53 accumulation. This
decreases its reactivity with the 2A10 monoclonal antibody. Phage display analysis is a
laboratory technique, for the study of proteins and protein-DNA that use bacteriophages to
connect proteins with the genetic information. From this analysis a consensus 2A10
recognition sequence was identified, possessing the core design DYS. Unexpectedly, this
design appears twice within the human Mdm2 molecule, at positions corresponding to
residues 258–260 and 393–395. Serine 395, residing within the carboxy-terminal 2A10
epitope, is the major target on Mdm2 for phosphorylation by ATM in vitro. (Ruth Maya et al,
2001)
Alpha-Msh is a a-melanocyte- stimulating hormone secreted by keratinocytes. Alpha-MSH
and other bioactive peptides are products of pro-opiomelanocortin (POMC). There is
biochemical and genetic evidence demonstrating that UV induction of POMC/MSH in skin is
directly controlled by p53. Whereas p53 potently stimulates the POMC promoter in
response to UV, the absence of p53 is associated with absence of the UV-tanning response.
The same pathway produces b-endorphin, another POMC derivative, which can contributes
to sun-seeking behavior. Furthermore, several instances of UV-independent pathologic
pigmentation are shown to involve p53 ‘‘mimicking’’ the tanning response. Therefore p53
functions as a sensor/effector for UV pigmentation, which is an everyday
environmental exposure. This pathway is activated in numerous conditions of
pathologic pigmentation and mimics the tanning response. (Rutao Cui et al. 2007)
Molecular aspects of tanning
Tanning is understood as an increase in epidermal melanisation in skin following UV
radiation, but can have a further role in a host response that protects against further UV-
induced damage. Tanning is mainly seen in humans but has been known to be seen in other
mammals such as sharks. (Barbara A. Gilchrest, 2011 ) Melanocytes (MC) sit along the
epidermal basal layer, have a minimal role except for constitutive melanin production. They
are usually only activated after sun exposure. (Graeme Walker et al, 2008)
Coat color study in animals has been an important factor in the study of genes that control
pigmentation in humans. In both humans and animals fully differentiated MCs
(Melanocytes) can be divided into two major groups, those in the hair bulb, whose function
is mainly to produce pigment for hair color, and epidermal MCs, which supply the epidermis
with melanin and are involved in the tanning response. (Graeme Walker et al, 2008)
After an UVR exposure to the skin, basal keratinocytes proliferate (resulting in epidermal
hyperplasia), to replace apoptotic “sunburn cells” that express p53. (Graeme Walker et al,
2008)
UVR may cause pigmentation in two ways by affecting the skin melanocytes. The first which
can be called Intermediate pigment darkening, fades rapidly and is due to oxidation of
already existing melanin in the melanocytes. The second, called Delayed tanning, appears a
lot later as it increases melanogenesis, allowing the darkened skin to be evident a few days
after UVR exposure and may last up to a week. (Graeme Walker et al, 2008)
For a long time it has been appreciated that tanning is the skins main response in protection
against UV radiation, and against acute and chronic damage. What we would consider a
“good tan” at best offers a moderate degree of photoprotection. If we compare a lightly
pigmentented epidermis with darkly pigemted epidermis, the darkly pigmented epidermis
reduces skin UV damage by a factor of 4.
Skin darkening regulated by UV radiation is a biphasic process. The initial darkening is due to
redistributioing and molecular changes to already existing melanin pigments within
epidermis. The second phase is due to up-regulation in melanin synthesis and its transfer to
keratinocytes. (John D’Orazio et al, 2013) This can show that a number of changes occur in
the epidermis due to the tanning response including melanogenesis, which is the synthesis
of melanin – pigment in hair, eyes and skin color. (Barbara A. Gilchres, 2011) It is known that
P53 Factor and KITLG expression are expressed in keratinized cells are exposure to UV.
(Graeme Walker et al, 2008) It has been possible to prove that after 24 hours there is a 2-3
fold increase of nucleotide repair protiens, after UV radiation. Also research has discovered
that keratinocyte derived gene products are regulated by UV. (Barbara A. Gilchrest, 2011)
Researchers have been unable to discover how P53 regulates KITLG, but they have ideas
based on the fact that most paracrine released from keritized cells after UV exposure are
regulated by P53, and that KITLG has receptors for P53 allowing regulation. (Graeme Walker
et al, 2008) UV has many other effects on the skin, including induction of an immune-
tolerant or immunosuppressive state and production of vitamin D by direct conversion of 7-
dehydrocholesterol into vitamin D3. (John D’Orazio et al,2013)
Artificial tanning
Over the years the number of artificial tanning methods such as indoor tanning salons have
become more popular. In 1980 only 1% of the American population had ever used a sunbed,
whereas now a huge 25% have used, or use them regularly.
Indoor tanning salons are known to be poorly regulated in terms of the strength of UV
radiation, they can be up to 10 times stronger than the UV inflicted by the sun, making the
tanning beds an authentic carcinogenic instrument. It has been proven that tanning can be
addictive, leading to more frequent and intense UV exposure. Lifetime risk of skin
melanoma increases by 75%for sunbed users under the age of 35. Since the molecular
pathways in the skin that activate UV-induced tanning result from cellular and DNA damage
it can result in skin damage and carcinogenesis, it appears this is the same case for tanning
salons but appears to be more intense.
For business purposes the industry commercializes the UV health benefits, emphasizing the
Vitamin D production, naturally made by the skin through 7-dehydrocholesterol into
Vitamin D3, but research has suggested that every day sun exposure as well as Vitamin D
containing foods and supplements exceed the required vitamin D levels the body needs.
This means extra exposure through tanning salons is unnecessary to prevent diseases such
as rickets. Decreasing UV radiation exposure, both naturally from sunlight and artificially
from tanning bed use, may be the single best way to reduce incidence of melanoma and
other skin cancers. (John D’Orazio et al, 2013)
